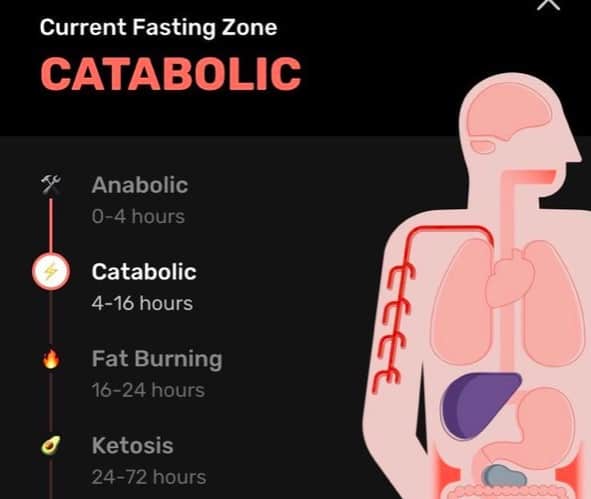 
That’s where the theory oversimplifies the science.Ĭhanges in muscle size depend on myofibrillar proteins. It’s also assumed that this will increase muscle mass by increasing NBAL. Glycogen provides energy for your muscles.Īfter exercising, it might seem logical to immediately eat protein and carbs to suppress MPB. Carb intake also inhibits MPB and aids glycogen resynthesis. Protein intake limits MPB and supports MPS. Post-workout nutrition can affect these processes. This balance is called net muscle protein balance (NBAL). The balance between MPB and MPS determines muscle growth. Muscle protein synthesis (MPS) also increases, but to a greater extent. 
According to a 2018 study, muscle protein breakdown (MPB) increases in response to strength training. It also claims that immediately eating protein and carbs is critical for: These processes are fueled by protein and carbs.Īccording to the anabolic state theory, this anabolic response is a limited time frame of only 30 minutes. This involves a range of cellular processes that facilitate muscle repair and growth. It’s the opposite of catabolism, or when larger molecules break down.Īfter strength training, your body is in an anabolic state. These molecules form into new cells and tissues, including muscle. This is the first report on an extracellular (alkaline) proteinase activity possibly serving a specific function in embryo implantation in a marsupial.The anabolic window theory is based on your body’s anabolic response.Īnabolism is when small molecules grow into bigger, complex molecules. The activity is correlated with the process of shedding of the extracellular embryo coverings (shell membrane) and of subsequent attachment of the trophoblast to the uterine epithelium, in the bilaminar but not the trilaminar (vascular) yolk-sac region. This enzyme appears to be extruded into the interface between trophoblast and uterine epithelium where it shows maximal activity for only approximately one day, around day (18-)19, exclusively in the bilaminar (avascular) yolk sac. A particularly interesting proteinase active at alkaline pH values is detected in the trophoblast-endoderm complex. Peak activity is found in the avascular (bilaminar) yolk sac at the phase when contact with the endometrium is being established. This enzyme is proposed to be a cathepsin indicating high catabolic activity connected particularly with protein transport from the endometrium into the yolk sac. At acid pH values an intracellular proteinase is detected in yolk sac endoderm and trophoblast as well as in endometrial glands and certain stromal cells. Proteinase patterns were correlated with light-microscopical morphology of the processes of shedding of the extracellular embryo coverings (shell membrane) and attachment of the trophoblast to the uterine epithelium. Proteinase patterns were studied using a sensitive histochemical gelatin-substrate-film test previously optimized for the detection of trophoblast-dependent proteinase (blastolemmase) in the rabbit. Tammar uteri with embryos were collected at diapause and at days 7, 17, 18, 19, 20, 21 and 26 of the 27-day gestational period. Since in eutherian mammals proteinases appear to play a central role in implantation-initiation mechanisms, a systematic histochemical investigation of proteinase patterns as related to implantation was performed in the tammar wallaby, Macropus eugenii (Macropodidae). Embryo implantation remains superficial (epithelio-chorial type) in most marsupials including the Macropodidae, but does involve formation of specialized contact zones of the trophoblast with the uterine epithelium.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
